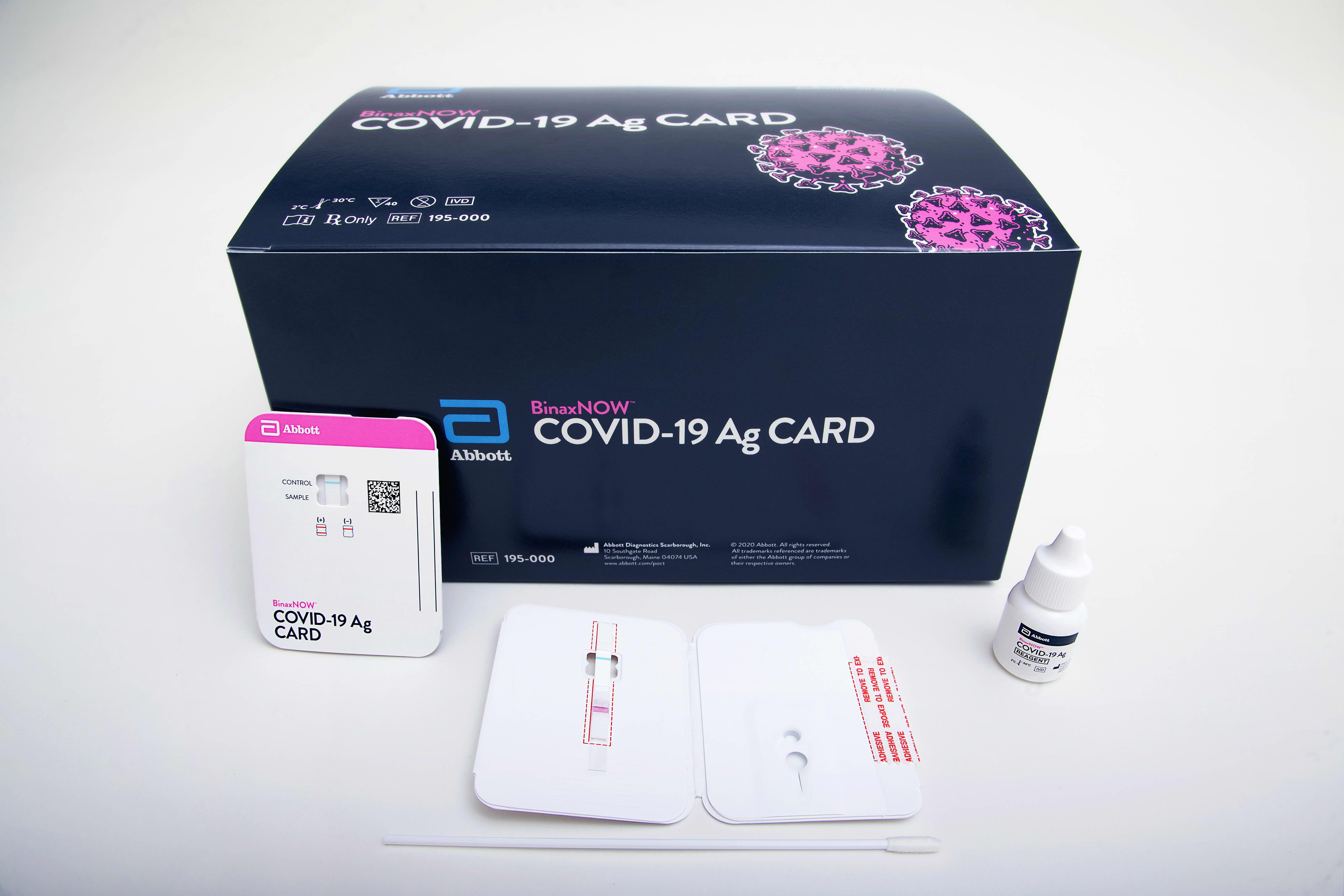
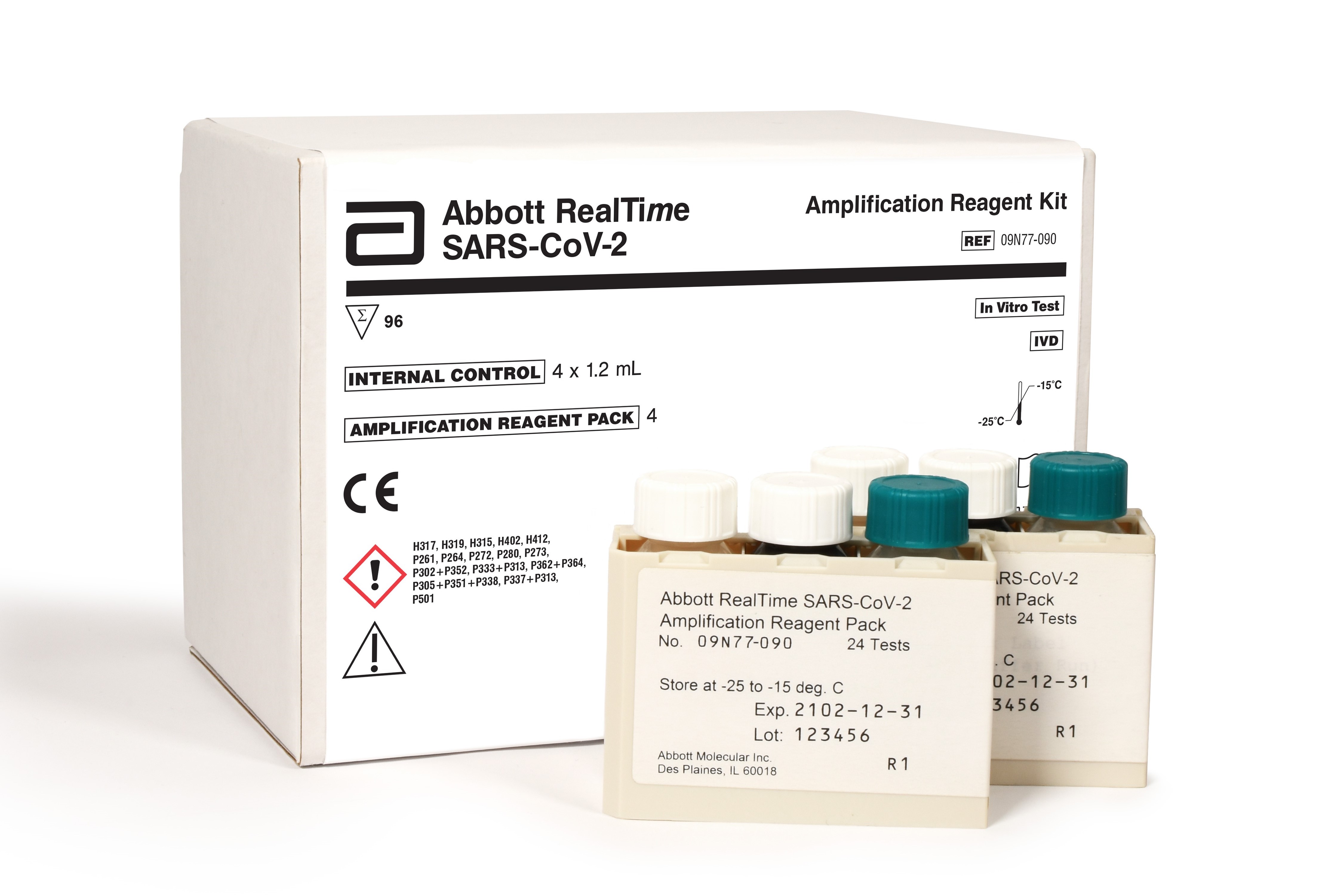
Abbott Labs is developing new coronavirus test for mass screening as US tries to reopen the economy; shares rise

12-17-Abbott-BinaxNOWHometest-packaging - Hamilton County Public Health | Hamilton County Public Health
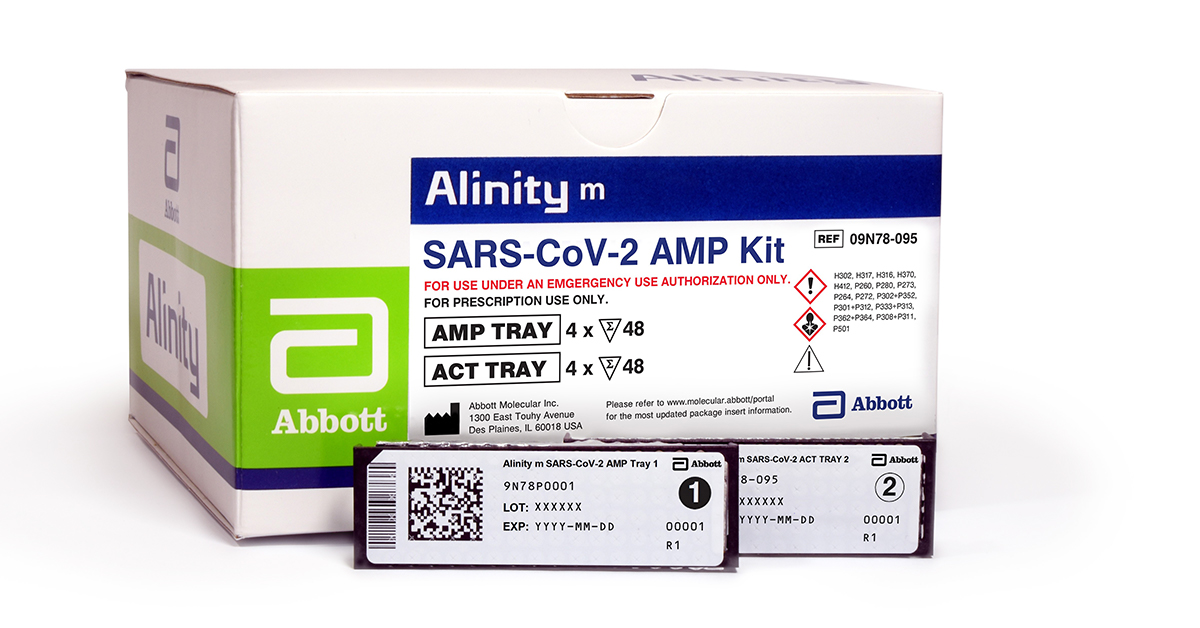
Abbott on Twitter: "ANNOUNCING: We've received FDA Emergency Use Authorization for our COVID-19 molecular test to be used on our new Alinity m System. What does this mean? Labs can run more