SciELO - Brasil - Comparative evaluation of the pH of calcium hydroxide powder in contact with carbon dioxide (CO2) Comparative evaluation of the pH of calcium hydroxide powder in contact with carbon

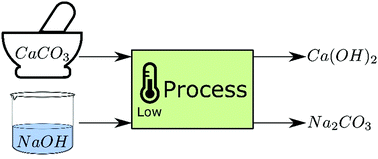
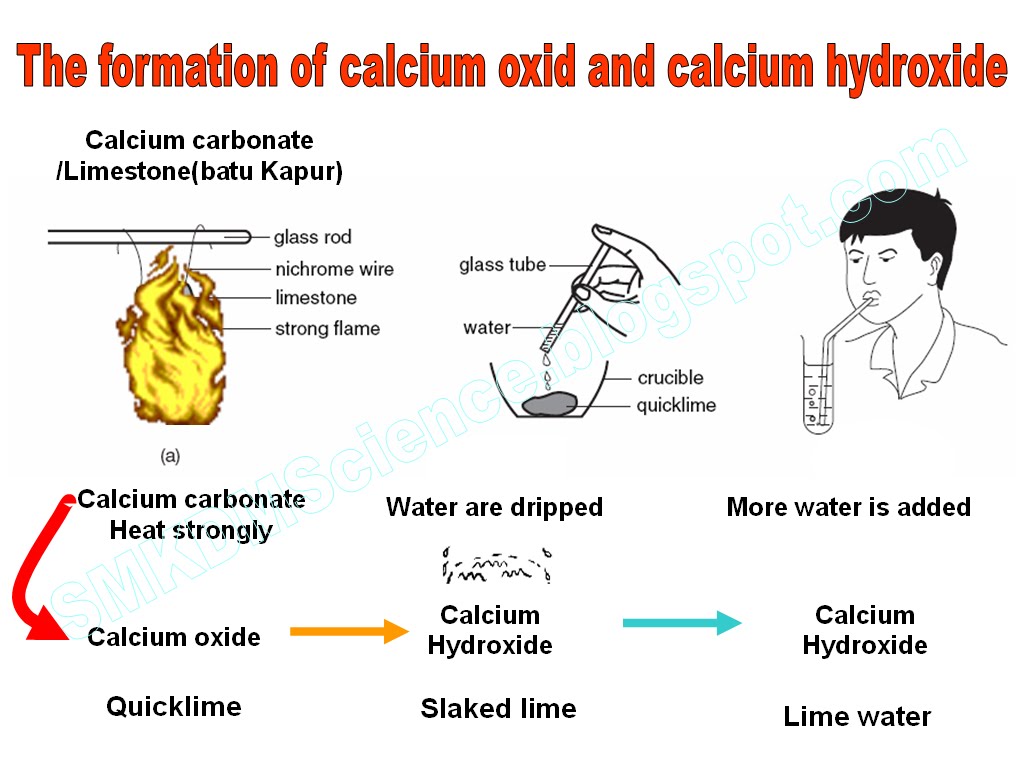
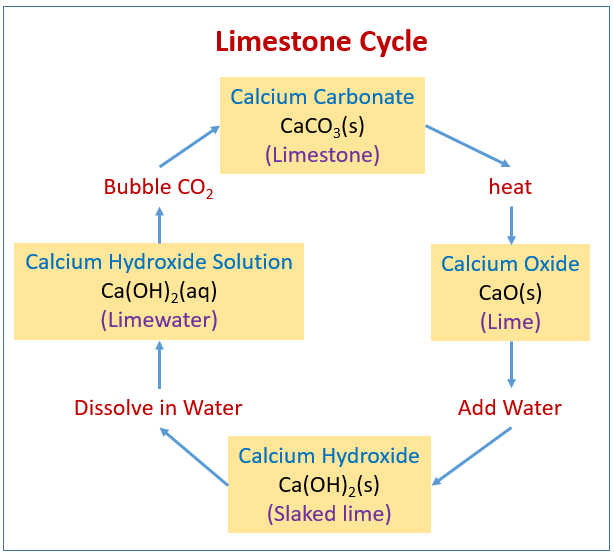
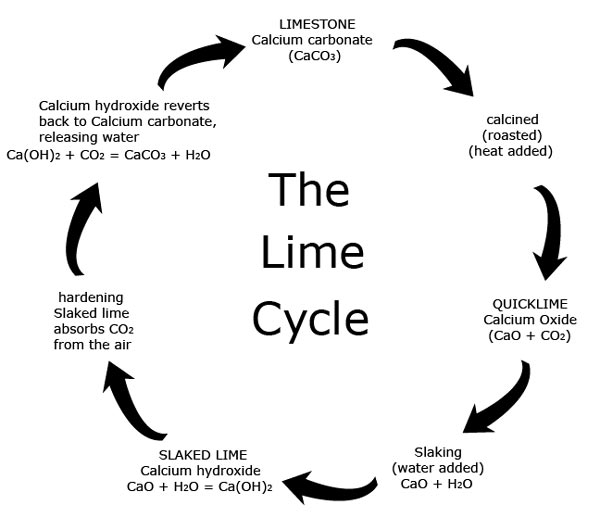
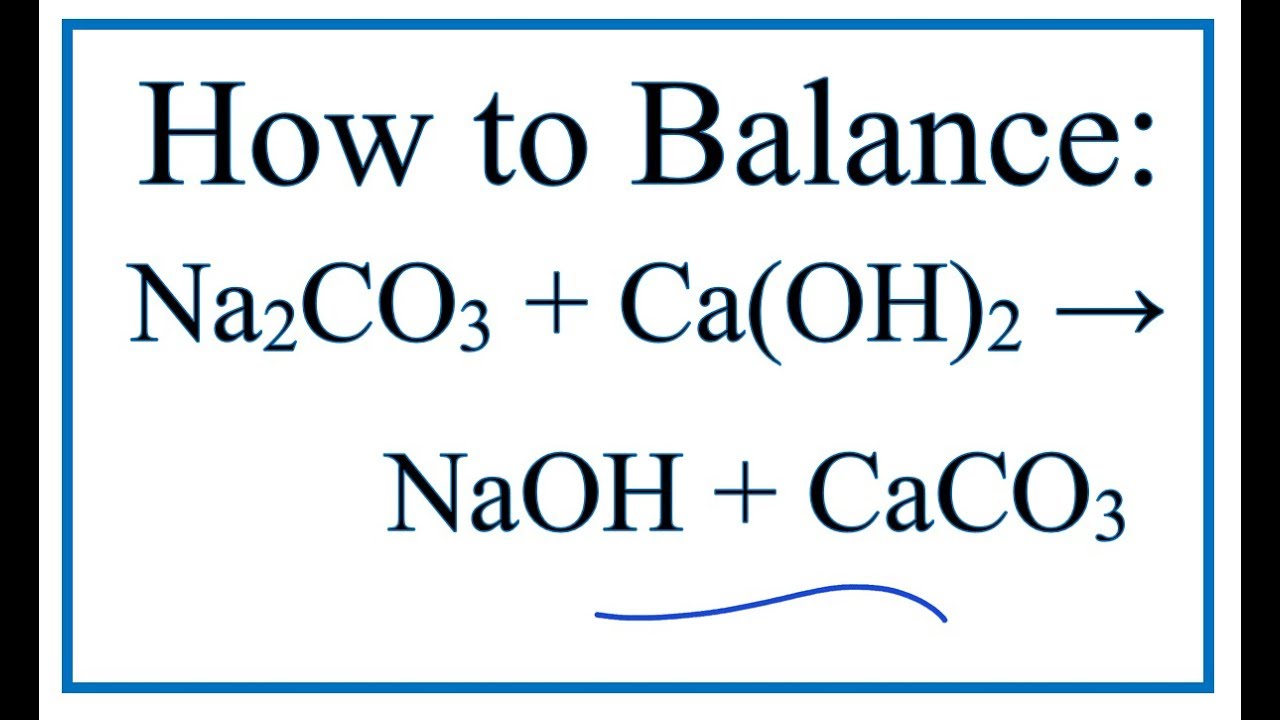
Write the balanced chemical equations for the following reactions.A Calcium hydroxide + Carbon dioxide → Calcium carbonate + waterB Zinc + Silver nitrate → Zinc nitrate + SilverC Aluminium + copper chloride

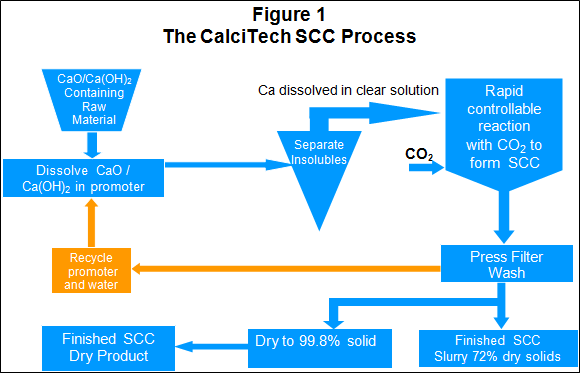
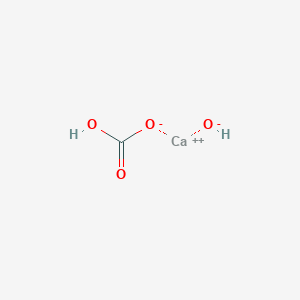
Novel one-step synthesis and characterization of bone-like carbonated apatite from calcium carbonate, calcium hydroxide and orthophosphoric acid as economical starting materials - ScienceDirect

Write the balanced chemical equation for the following reaction 1) Calcium Hydroxide + carbon dioxide - Brainly.in

Hydroxide + carbon dioxide is equal to calcium carbonate + water balance the equation numerically - Science - - 12540547 | Meritnation.com
Write the balanced chemical equations for the following reactions: i. Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water - Sarthaks eConnect | Largest Online Education Community

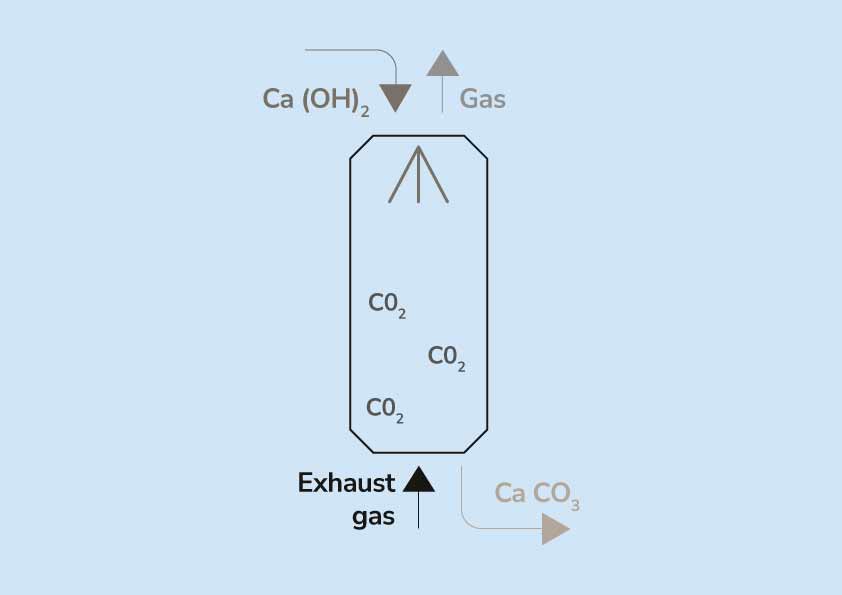
Distribution ofpH, calcium hydroxide, and calcium carbonate under the... | Download Scientific Diagram

Write the balance chemical equation for the following reactionsCalcium hydroxide + carbon dioxide → Calcium carbonate + WaterZinc + silver nitrate → Zinc nitrate + SilverAluminium + Copper chloride → Aluminium Chloride + Copper

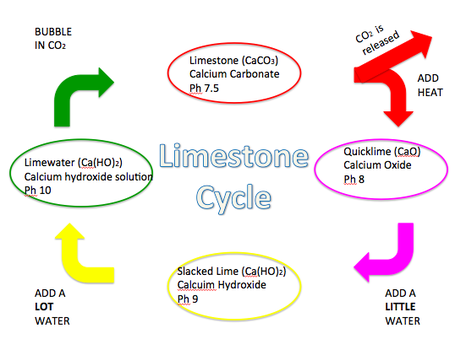
Difference Between Calcium Carbonate and Calcium Oxide | Compare the Difference Between Similar Terms