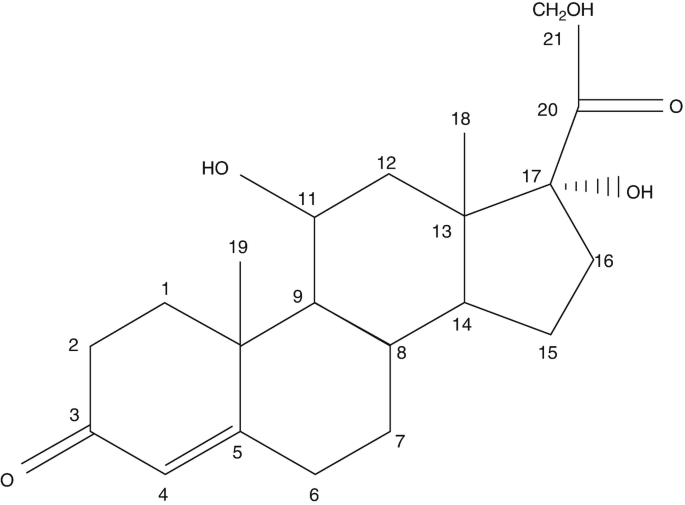
Edgar V. Lerma 🇵🇭 on Twitter: "@Nephro_Sparks Vitamin D analogs #CKDMBD ☝🏼 Note to self: Include equivalent dosing in 5th Edition #Nephpearls #NephSecrets #AskRenal https://t.co/JIN7QbFWWa" / Twitter

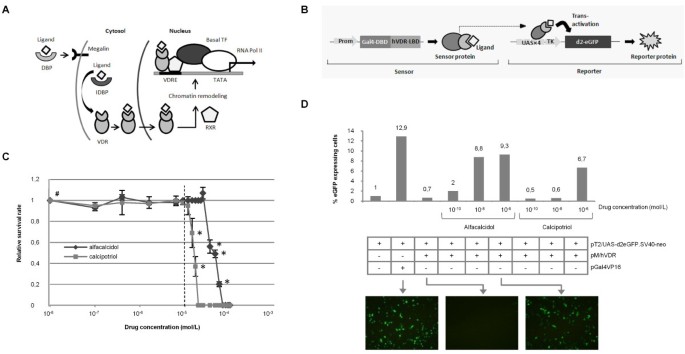
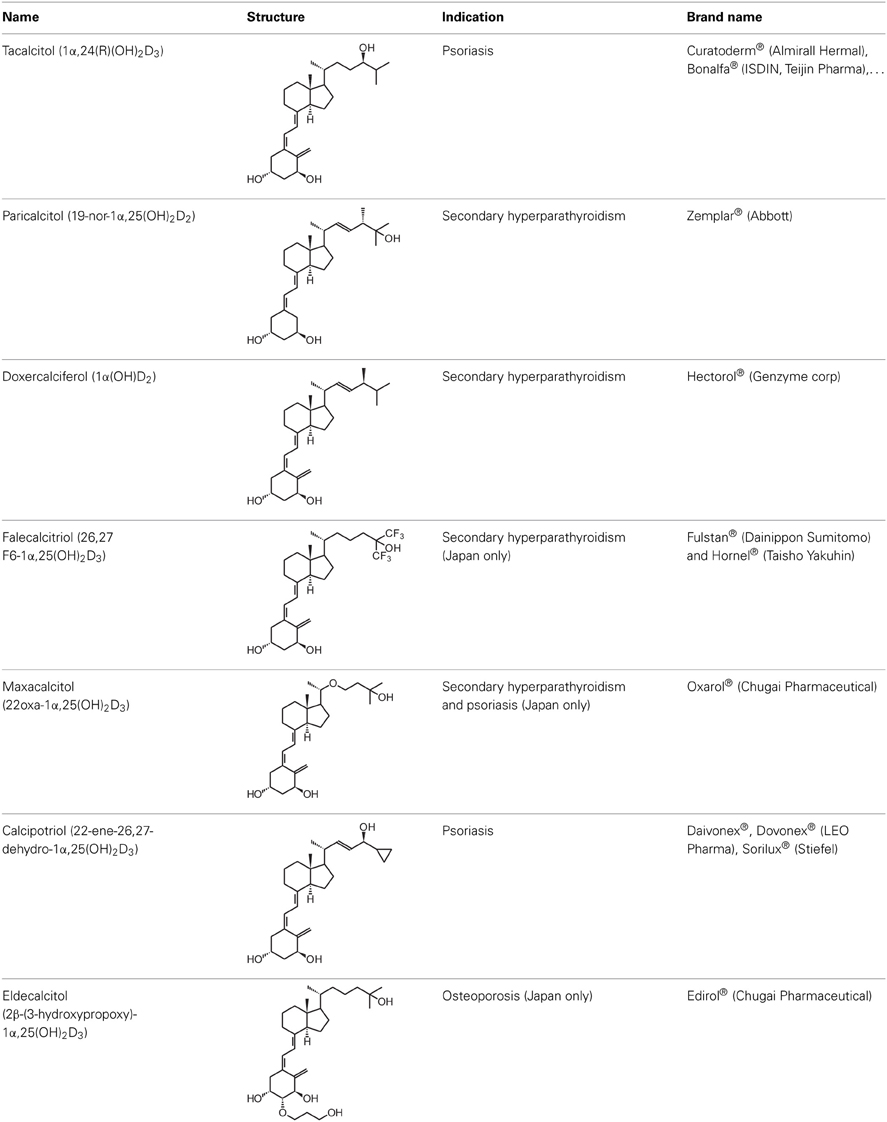
Drug Insight: vitamin D analogs in the treatment of secondary hyperparathyroidism in patients with chronic kidney disease | Nature Reviews Endocrinology

Synthesis and biological evaluation of all A-ring stereoisomers of 5,6-trans-2-methyl-1,25-dihydroxyvitamin D3 and their 20-epimers: possible binding modes of potent A-ring analogues to vitamin D receptor: Chemistry & Biology
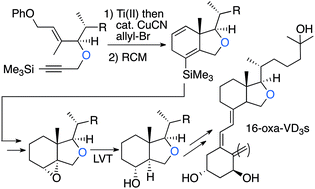
Synthesis and vitamin D receptor affinity of 16-oxa vitamin D3 analogues - Organic & Biomolecular Chemistry (RSC Publishing)
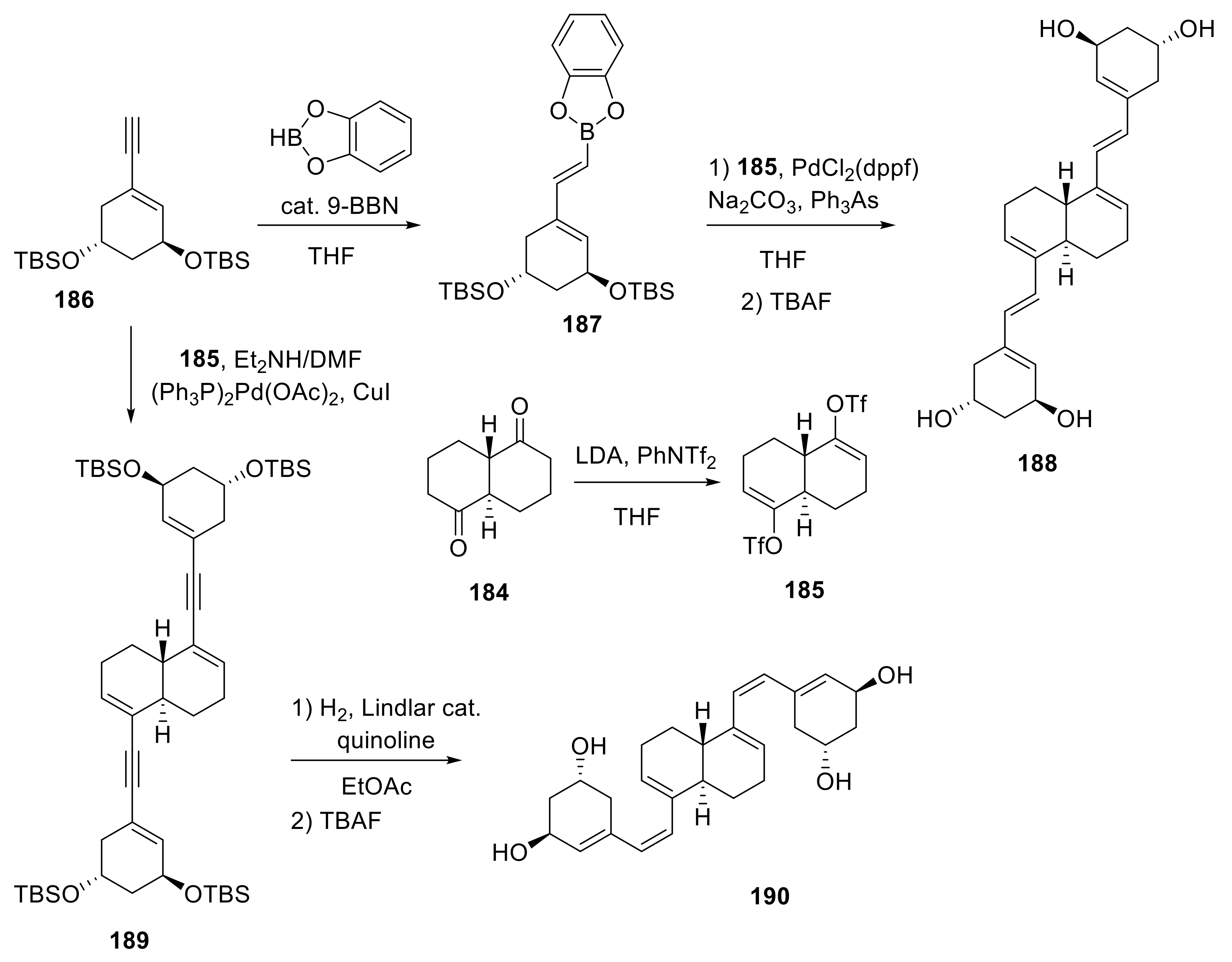
Biomolecules | Free Full-Text | The Synthesis and Biological Evaluation of D-Ring-Modified Vitamin D Analogues

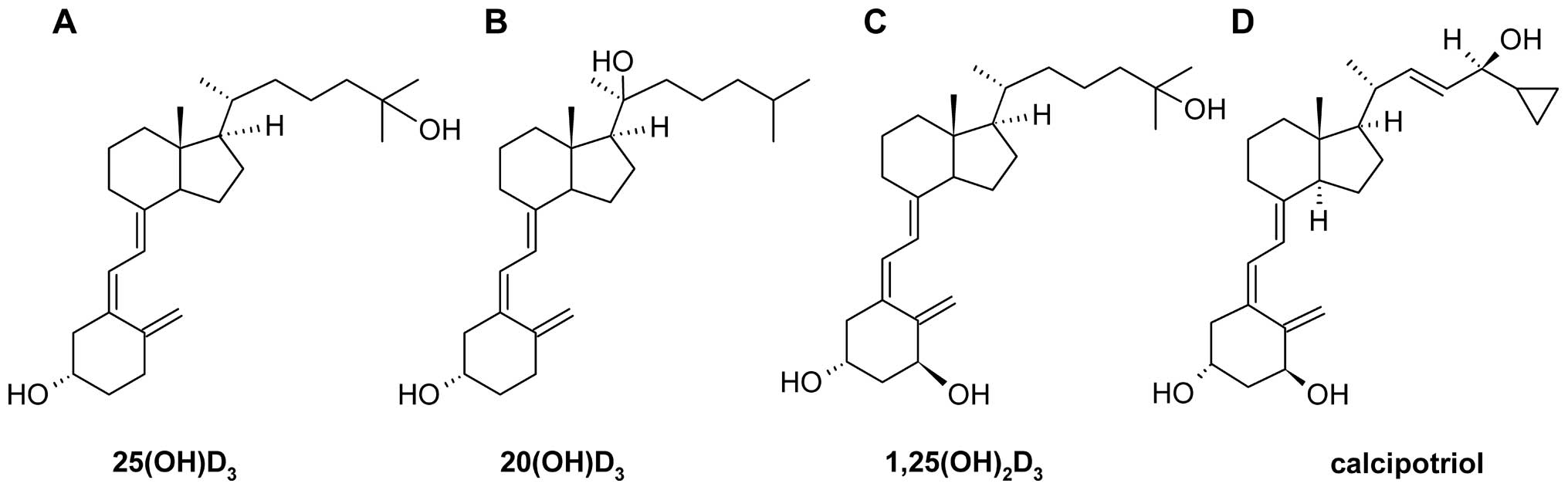
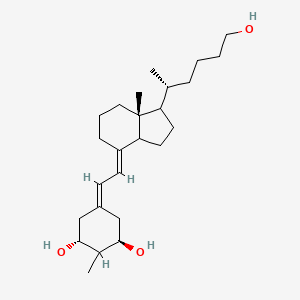
Different types of active vitamin D analogs including non-selective... | Download Scientific Diagram
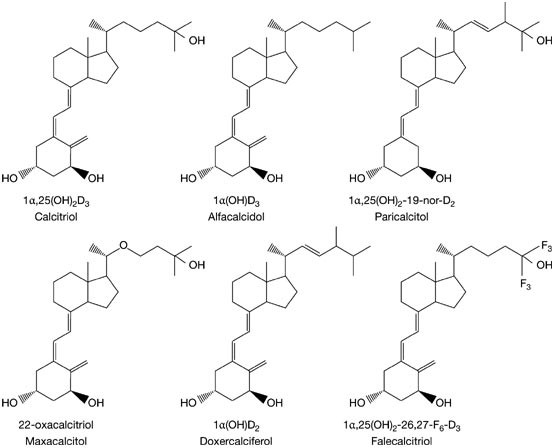
Drug Insight: vitamin D analogs in the treatment of secondary hyperparathyroidism in patients with chronic kidney disease | Nature Reviews Endocrinology